Bob Cowen and Jessica Luo got to present the concept of the Plankton portal to the oceanography community at the 2014 Ocean Science Meeting in Honolulu, Hawaii!
Category Archives: Science
Tree Map update and budding siphonophores
Thanks everyone for your phenomenal response to the treemap that I posted for the 300,000 classifications post. It’s really neat to be able to see how different users have classified more, or less over time. Of course, if you are one of the top classifiers, it’s fun to see your name up there!
At the suggestion of Lee Henderson, MD (wow! cool to see people from all professions and walks of life on Plankton Portal), I made an updated treemap with classifications from the past month. If there’s interest in periodic updates in this manner, let me know in the comments and I can post one of these up every couple weeks. But I don’t want to make it too much of a competition. Actually, it probably is a giant competition. So compete away!
Top 10 classifiers from the past month:
1. yshish
2. AnnaThema
3. phf13
4. Siiw
5. aplincoln
6. scopedriver
7. RachaelB
8. kredman
9. isadora paradijsvogel
10. ssushi
Thanks everyone!
On another note, our star moderator yshish emailed me today with a few questions about some cool siphonophores she found on PP. (side note: yshish has the BEST image collections on Talk. I regularly will be able to find specific images or taxa that I’m looking for by going through her collections.) Anyway, check out this series of images:
http://talk.planktonportal.org/#/subjects/APK00064cn 
http://talk.planktonportal.org/#/subjects/APK0003nz4  http://talk.planktonportal.org/#/subjects/APK0003xhv
http://talk.planktonportal.org/#/subjects/APK0003xhv 
You might remember that we featured the top image in a previous Fantastic Finds Friday (FFF) post. Now we bring it back because of the other two images that were found (the last one found 1 hr ago). These images found three months apart might be the same organism but in neighboring frames. We can’t tell right away, but once we go back into the raw data, we will be able to pull out the locations and times of these three images to check.
Now, the interesting question posed here is — is this siphonophore budding? Are these siphonophores asexually reproducing as we imaged it? And the answer — yes! absolutely! (I addressed this question earlier in a Talk post but now’s the time to feature it in the blogs!) All the little round bells that you see on the tentacles are small siphonophores developing from the gonozooids (reproductive ‘organs’) that will eventually be released and form free-swimming “eudoxids.” When they are released, these eudoxids will develop and then be capable of sexual reproduction. Weird, right? But wait, there’s more.
These eudoxids then develop little reproductive organs along its stem. Instead of eudoxids functionally male or female, they actually develop male and female reproductive parts – eggs and sperm – alternating between the two, sometimes regularly, sometimes irregularly. That way, they can ensure that the eggs are fertilized and can develop into larvae, then post-larvae, then adults.
The open ocean is a vast place, and animals have developed vastly different strategies for how to ensure the continuation of their species, whether it is in spawning aggregations (e.g. Grouper fish spawning in the Caribbean – and larvaceans also apparently form spawning aggregations) or being hermaphroditic (like Ctenophores and some fish), and being able to asexually bud and reproduce sexually. These siphonophores have adopted the strategy of being both hermaphroditic AND able to reproduce sexually and asexually.
This is the best diagram I’ve found to describe the life history of a Calycophoran siphonophore (all of the ‘rocketship’ and ‘two-cup’ siphonophores). This one is from C. Carre and D. Carre (1991).
Arrow worms: voracious plankton predators
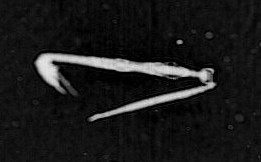
You may think orcas or great white sharks are the most voracious predators in the oceans, but based on their abundance and ability to consume a wide range of prey items, chaetognaths (a.k.a. “arrow worms”) give those big animals a run for their money. Large predators like sharks are extremely rare, but scoop up a bucket of seawater almost anywhere in the world and you are likely to find a few chaetognaths (if you have a microscope handy). Chaetognaths are transparent worms that often remain motionless in the water column, apparently relying on the element of surprise to capture a wide variety of plankton, including copepods, appendicularians, small fish larvae, and smaller chaetognaths. Chaetognaths are thought to be generalist feeders because their stomach contents often reflect the community captured by plankton nets. They use a mass of chitinous hooks around their mouths to capture prey – which gives them their name (“chaetognath” translates from Latin to mean “hairy jaw”) and a notoriously menacing appearance.

Chaetognaths are often straight in the ISIIS images but can also swim rapidly for short distances. The camera typically cannot resolve the tiny chitinous hooks on the chaetognath’s mouth.
Chaetognaths comprise about 100 species that are all typically 1-2 cm long. They are most abundant along the coasts, with some species being so sensitive to salinity that oceanographers can identify discrete water masses based solely on the community of chaetognath species. Similar to many other types of zooplankton, chaetognaths are hermaphrodites, first being male then changing into female as they get larger. Fertilized eggs can be attached to vegetation or encased in a gelatinous web. Eggs then hatch into juvenile chaetognaths, and thus they have no larval stage. This is called direct development because there is no process of metamorphosis.
The chaetognath’s body is streamlined and adapted to feeding with minimal visual input. The have sensory cilia that can detect small vibrations in the water that tell the chaetognaths that prey is within striking distance. With a quick flick of its tail, the chaetognath surges forward to capture the prey in its chitinous hooks used for grasping. It then transfers the prey to its mouth where it is swallowed whole. Some deeper water chaetognaths (>700 m deep) can even use bioluminescence to create a cloud of light that scientists think can be used to escape predation (Haddock and Case 1994).
References:
Haddock SHD and Case JF (1994) A bioluminescent chaetognath. Nature 367:225
Johnson WS and Allen DM (2005) Zooplankton of the Atlantic and Gulf coasts: A guide to the identification and ecology. Johns Hopkins University Press, Baltimore, MD
Lalli CM and Parsons TR (1997) Biological oceanography an introduction. Elsevier Butterworth-Heinemann, Burlington, MA
Jellyfish blooms, Norway and Periphylla
Today’s post comes from a new blog started by Andrea Bozman from the University of Nordland, Bodø, Norway. She is a Ph.D student studying the Helmet Jellyfish, Periphylla periphylla. From Andrea’s blog, www.smackofscience.com, she weighs in on the question of whether jellyfish blooms are increasing. In Norway and in many other areas of the world, people are worried that menace jellyfish might be eating all the young fish, devastating vital fisheries. And increasing jellyfish blooms only make matters worse. Is this the case? Actually, the jury is still out — some scientists are saying that jellyfish blooms are natural and just a part of a global cycle, while others say that jellyfish are increasing due to human-caused degradation of the marine environment. This kind of debate is healthy in science, and the conversation worth following.
Here chimes in Andrea.

Andrea Bozman from The University of Nordland, Bodø, Norway
Revenge of the blobs
Jellies have made a name for themselves in the news, without much effort on their part. Numerous stories linking jellies with negative events are reported world over. In tropical locations this is normally associated with stingers, those jellies that can prove fatal upon contact. Here in Norway, we are not exposed to such angry jellies, but their less hurtful relatives can have economic consequences for industries such as aquaculture and fisheries. In 2011 an influx of jellies in Kaldfjorden sunk a salmon net cage, resulting in an approximate 30 tonne loss of fish. However, the presence of jellies is not a new problem for Norwegian waters. We have our own contender that is rather happy up here – Periphylla periphylla.

Periphylla periphylla, a coronate jellyfish capable of living at depths of 200 – 2000 m. Photo credit: Erling Svensen/savethehighseas.org
The jelly juxtapose
Interactions between fish and jellyfish are not always a bad thing. Some jellies are known to eat fish, others are eaten by fish, and still others provide protection for young fish. Instead of assuming negative connotations on the easiest target – the soft-bodied blobs floating around in our seas – we need to conduct thorough studies on the systems. The increase in reports of jellies may be a compounded effect of media interest rather than an increase in actual jelly numbers. Jelly blooms are nothing new. Well preserved blooms have been found in the fossil record. That said, a change in the location and numbers of some jellies is a fact. And Periphylla in Norway is a prime example.
Yet like jelly blooms, the Periphylla story is also complex. Periphylla is one of the most globally distributed jellies and has always been in Norway. Why numbers are increasing in some areas is unclear. The occurrences may be new or may be part of a cycle. Periphylla is long lived and it may take years for an individual to reach the adult stage. The recent increased numbers in some fjords may have been years in the making. Our knowledge of jellies needs to grow as jellies are likely both misunderstood and underestimated players in the fjords.
Check out the whole blogpost at: http://smackofscience.com/2013/11/15/the-good-the-bad-and-the-jelly/
Thanks for continuing to help out with Plankton Portal! Help from volunteers like you contribute to our understanding of the life histories, distributions and behavior of jellyfish. This information is crucial to have to better understand how jellyfish blooms happen and whether they are increasing globally.
The Plankton (What does the Plankton say?)
Plankton Portal “en Français” (in french) coming soon!
Great news! We are working on translating Plankton Portal in French with our
French Collaborators: Fabrice Not from the Station Biologique de Roscoff and Jean-Olivier Irisson from the Observatoire Océanologique and Station Zoologique de Villefranche-sur-mer. “The idea is also to get some interest from French schools to develop a curriculum around Plankton Portal” Dr. Irisson explains. Stay Tuned.
Copepods: Rice of the Sea
Of all plankton groups, probably most is known about the copepods. They represent a critical link in the food chain and are consumed by diverse animal community ranging in size from small fish, chaetognaths, and ctenophores all the way up to large whales (the right whale is a voracious copepod feeder). Because of their small size and importance as food, copepods are affectionately known as “the rice of the sea.” Copepods are effectively captured by plankton nets because they have hard exoskeletons, and scientists have good estimates of their abundances and distributions. Although copepods are all relatively small (0.5 mm – 5 mm in length), they comprise over 200 families and 10,000 different species.

Examples of typical copepods. Note to the two large appendages on the top of the head with small sensory hairs
Copepods consume both phytoplankton and microzooplankton in two different ways: suspension feeding and raptorial feeding. Suspension feeding is relatively passive and performed by beating small appendages that draw a current through a feeding chamber. Copepods then select which particles encountered are food and discard others. Raptorial feeding is used to actively capture prey. Many copepods have small sensors on their first appendages to detect water disturbances produced by prey and also predators. They can use these relatively large appendages to “hop” through the water and capture an unsuspecting prey item or to quickly escape a predator.
Copepod reproductive strategies vary greatly and are adapted towards the ability to withstand the variable conditions that characterize the ocean environment. For example, many copepod eggs have the ability to enter a phase of diapause where they remain viable on the bottom for several months or even years, only hatching with conditions are favorable (high concentrations of food). Some copepods carry their eggs, allowing them to develop a bit before releasing them into the water column. The timing of copepod reproduction is especially important for the life cycle of fishes because most fish larvae depend on the recently hatched copepod nauplii for food. If there are not enough copepod nauplii present when fish larvae are abundant, there could be mass starvation events causing few fish larvae to reach their juvenile stage. Because of this, the copepod life cycle is extremely important to fish populations and overall ocean ecosystem health.

This image was taken from a thin layer near Stellwagen Bank offshore of Massachusetts, USA. Each one of the white particles is a copepod. The concentration of organisms in this image corresponds to ~400,000 individuals per cubic meter! That is some good eating for a right whale!
One of the most remarkable characteristics of copepods is their tendency to aggregate in discrete thin layers within the water column. Sometimes >90% of the copepod biomass will be confined these thin layers, which are a maximum of 5 m thick. ISIIS and other systems that sample on small scales are ideal for detecting these layers of copepods, and the function of the formation and dissipation of copepod thin layers is not well understood. Copepods have been shown to be attracted to strong changes in current direction and speed, potentially allowing them to feed at a faster rate within these zones (Woodson et al. 2005). The changes in environmental variables associated with aggregations of copepods are of great interest to marine ecologists. With your help, we can better understand how these extremely important organisms are distributed throughout our oceans!
References:
Johnson WS, Allen DM (2005) Zooplankton of the Atlantic and Gulf coasts: A guide to their identification and ecology. Johns Hopkins University Press. Baltimore, MD.
Woodson CB, Webster DR, Weissburg MJ, Yen J (2005) Response of copepods to physical gradients associated with structure in the ocean. Limnol Oceanogr 50:1552-1564
Pteropods By Dorothy Tang
Pteropods are a group of organisms that we’re not focusing on because they are not very abundant in the Plankton Portal dataset. Nevertheless, you may have run across a few of those fascinating little creatures.
Pteropod, which means ‘wing-foot’ in Greek, is a group of free-swimming pelagic gastropods (snails). Officially, the word ‘pteropod’ is no longer used in taxonomy; it is a collective term which refers to two clades of gastropods—thecosome (shelled body) and gymnosome (naked body). Pteropods are quite unique because in order to adapt to life in the water column, their foot is modified into two wing-like flippers used for swimming. Their body size ranges from a few millimeters to several centimeters – so they’re easily imaged by ISIIS. They can be quite abundant in certain regions of the world’s oceans, and are typically found near surface waters.
The first group of pteropods, thecosomes, are also known as the sea butterflies. They have a pair of large ‘wings’ and swims by continually flapping them. Their body is encased in a delicate and translucent shell.The shell can be coiled, needle-like, triangular, and globed.
Thecosomes are omnivores. Their diet consists of diatoms, dinoflagellates, and zooplanktons such as copepods, tintinnids, and other gastropod larvae. They capture food by secreting a spherical mucus web several times larger than their body. Scientists believe that the use of the large size mucus web is to capture large, fast swimming prey, such as copepods. The web acts as a filter: particles that are too large for ingestion are removed. During feeding, the mucus web is suspended above the animal while the animal remains motionless below. Ciliary action draws back the web to the mouth and the whole web is ingested.
Thecosome reproductive biology is quite unusual. The animal first matures and functions as male. The male pteropod mates with another male and the sperm is stored until the animal changes into a female. When the animal turns into female; its male reproductive organs degenerate. The female lays fertilized floating egg mass that later hatch into swimming larvae (veliger).
When a thecosome dies, its shell sinks to the bottom of the sea and forms sediment called pteropod ooze. The shell is composed of aragonite, an unstable form of carbonate mineral. Anthropogenic ocean acidification is one of the challenges that pteropods face. The increase of anthropogenic carbon dioxide level in the atmosphere reduces pH and carbonate ion concentration in the ocean, thus decreasing the calcium carbonate saturation level. As a result, the production of biogenic carbonate becomes more difficult. Overall, they have a hard time secreting their protective shell because of ocean acidification.
The second group of pteropods, or gymnosomes, are more commonly known as sea angels. They have much smaller wings which appear as side lobes. They are more robust and lack a shell. Unlike their thecosome relatives, gymnosomes are carnivores. They are active hunters and exclusively prey on thecosome pteropods. A combination of hooks and a toothed radula are employed to extract the flesh from the thecosomes’ shells.
The reproductive anatomy of gymnosome pteropods is similar to thecosomes pteropods. The only difference: the male reproductive organs do not degenerate in females. Gymnosomes has two distinct larvae forms. Eggs are hatched into shelled veliger. The veliger metamorphoses into a shell-less polytrochous larvae. The polytrochous larvae are initially wingless and movement is achieve by three ciliary bands. They gradually grow wings and lose the ciliary bands as they become adults.
Here is a very nice video about Pteropods.
Plankton Chronicles Project by Christian Sardet, CNRS / Noe Sardet and Sharif Mirshak, Parafilms. See Plankton Chronicles interactive site: planktonchronicles.org
Related articles
- Acid Ocean trailer (video) (news-oceanacidification-icc.org)
- Ocean Acidification Threatens Tiny ‘Sea Butterflies’ (pbs.org)
- The Gorgeous Shapes of Sea Butterflies (blogs.smithsonianmag.com)
FFF special behavior
Hello everyone. We have a special “behavior” Fantastic Finds Friday (FFF) today! These frames were selected from your posts to illustrate the power of the human eye to detect rare and unusual phenomena. The frames selected here may not be the most beautiful you have seen so far, but the story behind them is fascinating and could not be told without the help of our citizen scientists.
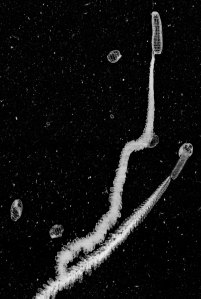
Here is great shot of a larvacean (also known as an appendicularian) getting spooked by the movement of ISIIS. Larvaceans are known to escape from their mucous house if threatened by a predator. Unfortunately the house can’t be used again, and they will start secreting a new house once the threat is passed.
Arrow worms (chaetognath) are voracious predators capable of engulfing prey as big as their own body. In these images, you can see an arrow worm catching a larvacean and the other grasping what appears to be a copepod. Their mouths resemble a crown of spikes ready to impale any unlucky prey. Chaetognaths also prey on fish larvae.
These two medusae just snagged a larvacean house. Accident or deliberate attempt to feed on these poor guys? The long trailing tentacles act like a sticky fishing net that retracts to bring in the catch of the day.
These Solmaris seem to be reaching for something (one tentacle pointed opposite to the others). Solmaris have been seen feeding on other jellies – even large siphonophores! They swim with their tentacles forward to maximize the chances of catching a prey. they then move the item to their mouth with one tentacle (like an arm almost).
No, these are really two different frames! Amazing consistency in posture isn’t it? And look at these two tentacles reaching out – sensing their environment? Hoping to encounter a tasty prey item? If we detect enough of these organisms, we could try to investigate at which time or location they behave this way. This could be a really interesting project!
So if you see something interesting like these example or suspect some interaction is at play in one of the frame use the hashtag #behavior. Remember to mark frames you want considered for future Fantastic Finds Friday posts with #FFF. Thanks, and keep up the good work!
Salps and Doliolids
Salps and doliolids (class Thaliacea) are interesting animals because they are in the phylum Chordata, which includes all animals with a notochord during development (e.g., humans, fish, cats), but thaliaceans have a vastly different appearance and feeding strategy compared to most vertebrates. A salp or doliolid body is essentially a giant pumping muscle that forces water through a mucous net filter that collects phytoplankton and is ingested periodically. Both groups have limited mobility, with salps using muscular contractions to scoot through the water, while doliolids use tiny beating cilia to propel themselves.
The life history of salps and doliolids is remarkable and complex. Similar to plants, their life cycle alternates between sexual and asexual generations. The solitary phase reproduces asexually by budding off clones of itself. On salps, a chain of these clones develops on the solitary animal that is then released and reproduces sexually with other salp chains. The chains first mature as female and then change sexes to become male when they are larger! These chains release small solitary salps that then begin asexual budding once they are a certain size. Doliolids on the other hand produce short-lived tadpole larvae that are not seen in salps. When you consider that a chain of salps contains an average of ~28 individuals, it is no surprise that these organisms are capable of extremely fast reproductive rates and can double their populations in hours (Heron 1972). Some scientists think their remarkable reproductive rates can overwhelm other phytoplankton grazers, which could explain the fact that large salp aggregations are often associated with low biomass of other grazers (Alldredge and Madin 1982).
Because of their ability to reproduce quickly, salps are often very abundant near steady supplies of phytoplankton, such as at ocean fronts (zones where two water masses with differing physical properties meet) and eddies (Deibel and Paffenhöfer 2009). However, these organisms cannot tolerate extremely dense aggregations of phytoplankton because their mucous filters will become clogged with prey, which severely decreases their feeding efficiency. Salps and doliolids can “bloom” like other jellies, and when these blooms die off the dead salp bodies can export a large amount of carbon into deeper waters. Because of salps and doliolids close evolutionary relationship to vertebrates, scientists are also very interested in their developmental biology. Scientists are trying to use salps as a model organism to study the development of complex nervous systems in all vertebrate animals (Lacalli and Holland 1998).
Check out this video from Plankton Chronicles on these remarkable animals!
Plankton Chronicles Project by Christian Sardet, CNRS / Noe Sardet and Sharif Mirshak, Parafilms. See Plankton Chronicles interactive site: planktonchronicles.org
References:
Alldredge AL and Madin LP (1982) Pelagic tunicates: Unique herbivores in the marine plankton. Bioscience 32:655-663
Deibel D and Paffenhöfer GA (2009) Predictability of patches of neritic salps and doliolids (tunicata, thaliacea). J Plankton Res 31:1571-1579
Heron AC (1972) Population ecology of a colonizing species: The pelagic tunicate Thalia democratica – I. individual growth rate and generation time. Oecologia 10:269-293
Lacalli TC and Holland LZ (1998) The developing dorsal ganglion of the salp Thalia democratica, and the nature of the ancestral chordate brain. Philosophical Transactions of the Royal Society B: Biological Sciences 353:1943-1967